
DePuy ASR & ASR-XL Models:
On August 24, 2010, the United States Food and Drug Administration recalled the DePuy ASR and ASR-XL hip implants. These defective products had been implanted in hip replacement patients since 2005, and they were recalled because one in eight recipients of the defective device needed surgery to replace or correct the implant within five years of receiving it. This is an extremely high rate of failure for a medical product, and is particularly concerning because patients had to undergo multiple extensive and painful surgeries and rehabilitation.
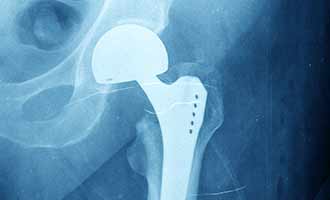 The implants are failing due to loosening and misalignment of components. The metal-on-metal design of the implants that are misaligned causes metal in the implant to flake off and enter the bloodstream of the person with the implant.
The implants are failing due to loosening and misalignment of components. The metal-on-metal design of the implants that are misaligned causes metal in the implant to flake off and enter the bloodstream of the person with the implant.
In addition to unnecessary, painful revision surgeries, recipients of these defective implants have experienced cobalt and chromium toxicity from metal in the bloodstream, which can cause vision impairment and hearing loss. The DePuy ASR hip implant can also cause infection in the area of the implant, fracture of the bone, dislocation of the joint, swelling, nerve damage, tissue damage, and muscle damage. If you or a loved one have been the recipient of the recalled ASR or ASR-XL hip implant manufactured by DePuy, you have the right to file a lawsuit against DePuy to seek compensation for your injuries, and the personal injury attorneys at The Law Offices of d’Oliveira & Associates can help you.

The DePuy Pinnacle Model:
DePuy Hip manufactures several different models of hip replacements. In addition to the recalled ASR and ASR-XL models, they also manufacture the Pinnacle model. The Pinnacle model has been on the market since 2002, and the ASR and ASR-XL models have been used since 2005.
Shortly after being introduced to the market, DePuy received complaints that the DePuy Pinnacle model was defective as early as 2002. Since then, many patients and doctors have reported problems with the DePuy Pinnacle model that are similar to the problems with the ASR and ASR-XL models.
There are concerns that the Pinnacle hip replacements may have failure rates similar to the ASR and ASR-XL models, which had failure rates of 13%, roughly 1 in 8 patients.
Although the DePuy Pinnacle model has not yet been recalled, many recipients of the DePuy Pinnacle implant have filed lawsuits against DePuy. With the ASR-and ASR-XL models, the FDA did not recall the defective product until one year after the first lawsuit against DePuy for the ASR models. Even though the Pinnacle model has not been recalled, if you have the Pinnacle implant, you may have the right to file a lawsuit against DePuy Hip Devices, and the personal injury attorneys at The Law Office of d’Oliveira & Associates can help you.
What to do if You Think You Have Received a DePuy Implant
The manufacturers of the defective hip implants are using aggressive tactics to contact implant recipients and try to convince them not to participate in a lawsuit against DePuy. This is soley to protect the financial interest of the DePuy company, who manufactured these defective products. If you or a loved one have experienced any pain from either a Pinnacle or ASR/ASR-XL hip replacement, call the Law Offices of d’Oliveira & Associates now, so that we can help make sure your interests are protected.
We can help you find a personal injury attorney to assist you in making important personal and legal decisions. A personal injury lawyer can help you assert your rights and get you the compensation that you deserve. Fill out a contact form or call us now at 1-800-992-6878 for FREE case evaluation. For your convenience, our telephones are answered 24 hours a day and 7 days a week.



