
Jury selection is expected to begin in early December for the next Risperdal trial in spite of jury awards or settlements in five of the six cases that have already gone to court. Attorneys for Risperdal manufacturers are saying that a class-wide settlement is not in the works, and they plan to move forward with the litigation. The most recent settlement came earlier this month when Janssen Pharmaceuticals, a subsidiary of Johnson & Johnson that manufacturers the antipsychotic drug, settled with the family of a boy who grew excessive breast tissue after taking Risperdal. The lawsuit was settled days before it was set to go to trial. Johnson & Johnson is currently facing thousands of Risperdal lawsuits alleging injuries as a result of taking the drug.
Will More of the Risperdal Lawsuits be Settled?
 According to court papers, the case involved a boy diagnosed with Asperger’s syndrome, impulse control disorder and oppositional defiance disorder, who was first prescribed Risperdal when he was 7. The plaintiff contended that Risperdal caused gynecomastia, a condition in which young men and boys grow excessive and permanent breast tissue and that Janssen did not warn consumers of these risks in spite of knowing about them. Janssen’s lawyers ultimately decided to settle instead of taking the case to trial, raising the question if more settlements may be made in the pending Risperdal litigation.
According to court papers, the case involved a boy diagnosed with Asperger’s syndrome, impulse control disorder and oppositional defiance disorder, who was first prescribed Risperdal when he was 7. The plaintiff contended that Risperdal caused gynecomastia, a condition in which young men and boys grow excessive and permanent breast tissue and that Janssen did not warn consumers of these risks in spite of knowing about them. Janssen’s lawyers ultimately decided to settle instead of taking the case to trial, raising the question if more settlements may be made in the pending Risperdal litigation.
The plaintiff’s lawyer said, however, that this was a “one-off” settlement and that there are “no global settlement talks underway” that would resolve the Risperdal mass tort program in Philadelphia. Despite major setbacks in many of the cases that have gone to trial, Johnson & Johnson’s litigation strategy remains unchanged, according to attorneys. The pharmaceutical company still refuses to accept responsibility for this dangerous drug that has caused adverse and devastating health effects in so many young children.
Is There Evidence Linking Risperdal to Gynecomastia?
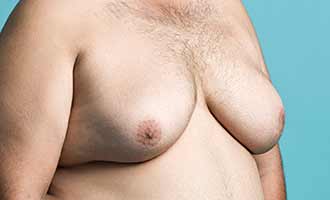 There have been a number of studies that have shown an increase in hormones related to breast tissue production in patients treated with Risperdal. These risks are more likely to affect young men and adolescent males who are still going through hormonal changes and developing physically. A study published in the November 2003 issue of The Journal of Clinical Psychiatry stated that long-term treatment with the drug in young boys and male adolescents significantly raised hormone levels. More recently, in March 2014, a team of doctors from the University of British Columbia conducted an independent drug safety review of Risperdal and its connection to breast enlargement in men and found that patients using the medication faced a 69 percent increased risk of gynecomastia.
There have been a number of studies that have shown an increase in hormones related to breast tissue production in patients treated with Risperdal. These risks are more likely to affect young men and adolescent males who are still going through hormonal changes and developing physically. A study published in the November 2003 issue of The Journal of Clinical Psychiatry stated that long-term treatment with the drug in young boys and male adolescents significantly raised hormone levels. More recently, in March 2014, a team of doctors from the University of British Columbia conducted an independent drug safety review of Risperdal and its connection to breast enlargement in men and found that patients using the medication faced a 69 percent increased risk of gynecomastia.
Contact an Experienced Risperdal Lawyer
 If you or someone you know has developed gynecomastia after taking the drug Risperdal, you may be able to receive compensation for your medical bills, lost income and pain and suffering, among other losses. Our firm is working with experienced Risperdal lawyers across the country, who are investigating these cases. They may be able to file a Risperdal lawsuit for you, and they charge no fee unless you win an award or settlement. For a free (no obligation) case evaluation, call our toll free number at 1-800-992-6878 or fill out a contact form online.
If you or someone you know has developed gynecomastia after taking the drug Risperdal, you may be able to receive compensation for your medical bills, lost income and pain and suffering, among other losses. Our firm is working with experienced Risperdal lawyers across the country, who are investigating these cases. They may be able to file a Risperdal lawsuit for you, and they charge no fee unless you win an award or settlement. For a free (no obligation) case evaluation, call our toll free number at 1-800-992-6878 or fill out a contact form online.
Source: http://www.thelegalintelligencer.com/id=1202771211269/Risperdal-Case-Settles-Before-Trial-Begins


