
If you have suffered serious side effects from Wright Profemur Hip Implant, you may be entitled to compensation for your medical bills, lost income, pain and suffering, among other losses. We are currently working with some of the most experienced MicroPort ProFemur Hip Implant lawyers in the country, who may file a lawsuit on your behalf. There is no legal fee collected until you obtain a settlement or award. For a free (no obligation) case evaluation fill out a contact form online or call us toll free at 1-800-992-6878.
What Is A MicroPort ProFemur Hip Implant?
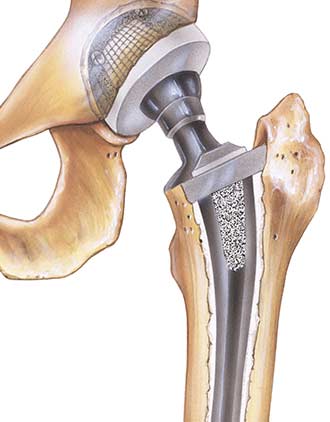 MicroPort Orthopedics, the producer of this ProFemur Hip Implant, began manufacturing the device after buying out Wright Medical Group’s OrthoRecon business for $290 million. Wright Medical Group, at the time, had been the leading company for the ProFemur device, and is now the subject of hundreds of ProFemur Hip Implant lawsuits throughout the country. This hip replacement device entails a metal-on-metal design to re-create natural hip movement for those who may be facing pain from walking or a deteriorating hip bone. This metal-on-metal design consists of a metal ball, connected to a metal hip stem, being placed within a metal socket to create a replica hip.
MicroPort Orthopedics, the producer of this ProFemur Hip Implant, began manufacturing the device after buying out Wright Medical Group’s OrthoRecon business for $290 million. Wright Medical Group, at the time, had been the leading company for the ProFemur device, and is now the subject of hundreds of ProFemur Hip Implant lawsuits throughout the country. This hip replacement device entails a metal-on-metal design to re-create natural hip movement for those who may be facing pain from walking or a deteriorating hip bone. This metal-on-metal design consists of a metal ball, connected to a metal hip stem, being placed within a metal socket to create a replica hip.
One of the preliminary advantages of using this type of hip replacement design was the ability to adjust the device to perfectly fit into the hip socket, allowing for an easier transition for patients. By designing this device in such a way, an increase in flexibility would allow for patients to begin walking much faster than those who choose to have another hip replacement design placed in the body. The average expected “life” of this device, as stated by the manufacturer, is 15 years.
What Is Dangerous About The MicroPort Profemur Hip Implant?
 According to a U.S. Food and Drug Administration, or FDA, Class 1 Recall, the ProFemur modular neck components of the hip replacement device are unexpectedly fracturing, causing instability and fierce pain. If a patients does not realize that their MicroPort ProFemur device is failing or fractured, serious consequences, including neurovascular damage, hemorrhaging, or even death can occur.
According to a U.S. Food and Drug Administration, or FDA, Class 1 Recall, the ProFemur modular neck components of the hip replacement device are unexpectedly fracturing, causing instability and fierce pain. If a patients does not realize that their MicroPort ProFemur device is failing or fractured, serious consequences, including neurovascular damage, hemorrhaging, or even death can occur.
What Side Effects Are Linked To The MicroPort Profemur Hip Implant?
- Severe inflammation
- Sudden pain
- Difficulty walking or standing
- Reduction in motion around the hip
- Redness
- Instability
Has The FDA Taken Action In Regards To These MicroPort ProFemur Hip Implant Complaints?
 In August 2015, the FDA issued a Class I recall for the ProFemur Neck Varus/Valgus CoCR (Part #PHAC1254). In total, more than 10,500 units, distributed between June 2009 and July 2015, were included in the recall. A Class I recall is the most-serious classes for medical devices, and the highest recall that the FDA may issue.
In August 2015, the FDA issued a Class I recall for the ProFemur Neck Varus/Valgus CoCR (Part #PHAC1254). In total, more than 10,500 units, distributed between June 2009 and July 2015, were included in the recall. A Class I recall is the most-serious classes for medical devices, and the highest recall that the FDA may issue.
MicroPort responded immediately to this Class I recall by notifying hospitals, doctors, surgeons and patients that those who have implanted or have been implanted with this modular neck portion of the hip replacement device will need revision surgery as soon as possible. The manufacturer went as far as stating in their own report that failing to have revision surgery could lead to death. Doctors and surgeons were ordered to halt any hip replacement surgeries that involved this particular part.
Why Contact d’Oliveira & Associates?
 If you or someone you know has suffered from serious MicroPort ProFemur Hip Implant side effects, you may be entitled to compensation. Our law firm is currently working with some of the more experienced MicroPort ProFemur Hip Implant lawyers in the country who are handling these cases and charge no legal fees unless you receive a settlement or award. Please contact our toll free number 24/7 at 1-800-992-6878, or fill out a contact form for a free (no obligation) case evaluation.
If you or someone you know has suffered from serious MicroPort ProFemur Hip Implant side effects, you may be entitled to compensation. Our law firm is currently working with some of the more experienced MicroPort ProFemur Hip Implant lawyers in the country who are handling these cases and charge no legal fees unless you receive a settlement or award. Please contact our toll free number 24/7 at 1-800-992-6878, or fill out a contact form for a free (no obligation) case evaluation.
